 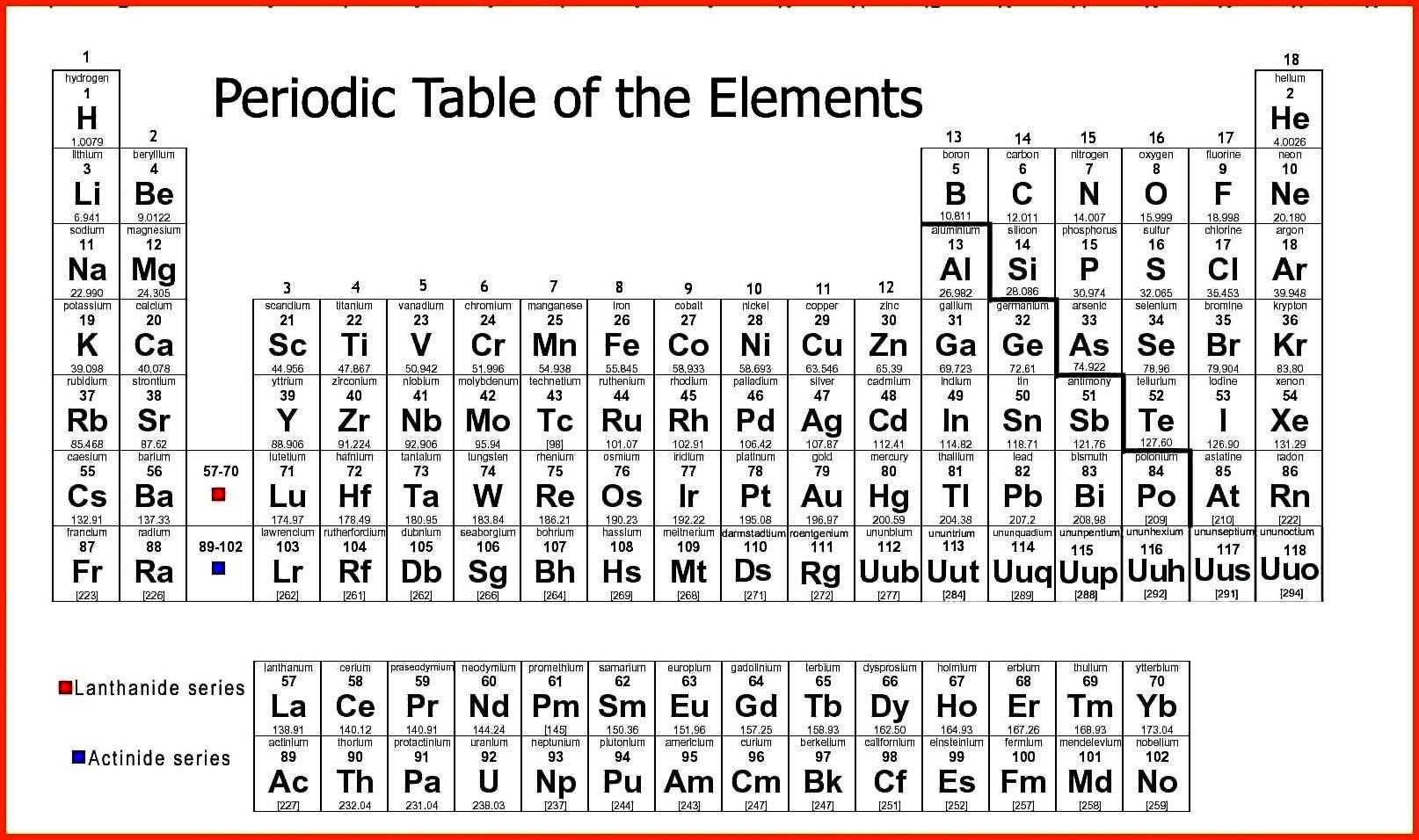 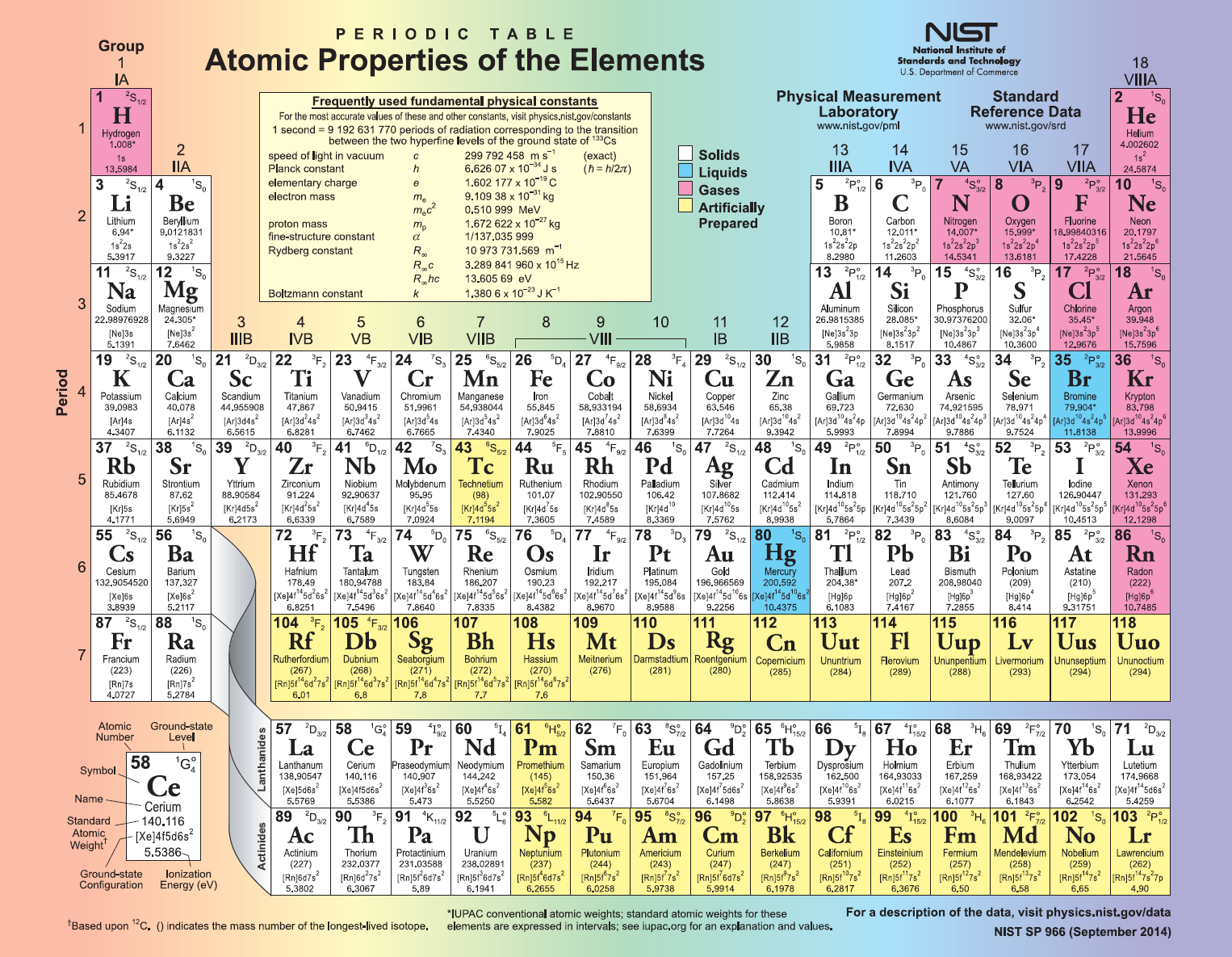
Mendeleev left gaps where there was no known element matching the anticipated weight and properties. Frequently incorrect weights were reported and later revised when purer samples could be analyzed. He was correct because many elements were difficult to isolate in the samples at the time. Mendeleev trusted experimental evidence of chemical reactions more than the measured weights. He used atomic weights to sort the elements however, he reordered them based on observed properties if they did not seem to be in the right spots. In 1863 Dmitri Mendeleev drafted the first of his 60 versions of the Periodic Table. He observed there were similar properties every eight elements, so he organized his table with eight columns. John Newlands organized elements based on weights. Let us take a brief look at both the history and the most common versions in use today. While the information about each element in the Periodic Table is the same, different formats have been used to organize the Periodic Table. Hmmm, is this element a Ravenclaw or Hufflepuff? Versions of the Periodic Table For Harry Potter fans, think of it as an algorithmic version of a sorting hat. The organization of the Periodic Table in Rows and Columns is not haphazard. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
